Biosimilar peptide (Liraglutide) for the treatment of Type 2 Diabetes
Type 2 diabetes is a chronic disease where tissues develop resistance to insulin. In this regard any insulin secreted by the pancreas cannot be utilized to absorb glucose. According to the International Diabetes Federation, 592 million people will suffer from type 2 diabetes by 2035.
Liraglutide, is an anti-diabetic medication used to for the treatment of type 2 diabetes, obesity, and for chronic weight management. Liraglutide is a glucagon-like peptide-1 receptor agonist (GLP-1 receptor agonist) also known as incretin mimetic. It works by increasing insulin release from the beta cells in the pancreas and decreases excessive glucagon release.
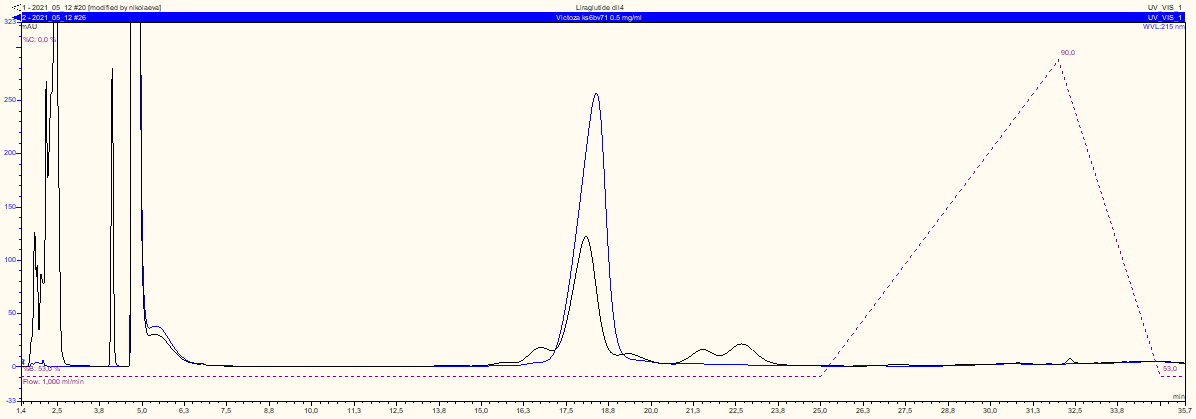
AVVA Pharmaceuticals is in the process of developing the Liraglutide Biosimilar for human use.
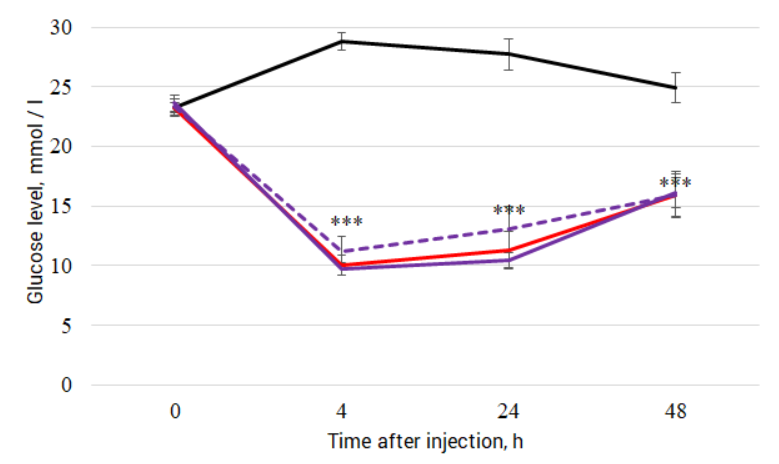
Effect of Victoza and Liraglutide AVVA tested in laboratory mice db/db. Injections of Victoza and Liraglutide AVVA at a concentration of 300 and 600 μg/kg led to a significant decrease in blood glucose from 4 to 48 hours after administration: F (3.40) = 20.18, p <0.001; F (3.40) = 62.04, p <0.001 and F (3.40) = 13.23, p <0.001, respectively. Blood glucose levels in male and female db / db mice after 12 hours fasting and subcutaneous injection. Saline solution (solid black line) Victoza (red line) Liraglutide AVVA 300 μg / kg (purple solid line) Liraglutide AVVA 600 μg / kg (purple dotted line). *** - differences from the control group LSD test p <0.001.
Inventor’s Team

Vasily N. Stepanenko, PhD
Head of Pilot Biotech Production at the Shemyakin and Ovchinnikov Institute of Bioorganic Chemistry